Important facts
- What is considered an intermediate product under the EUDR?
- Intermediate products are processed raw materials such as flour, cocoa mass or wooden boards that are produced before the end product and must remain traceable.
- When does a new EUDR obligation begin?
- A new duty of care arises as soon as a product is supplied commercially to another company, but not if it has already been placed on the EU market unchanged.
- How detailed must traceability be for collective batches?
- Each partial quantity of a collection batch must be clearly documented in terms of origin, parcel ID and quantity.
- Does all internal processing have to be documented?
- Detailed documentation of internal processing steps is only necessary in the event of mixing or increased risk.
- How can companies ensure EUDR-compliant traceability?
- Through digital systems, clear processes and risk-based documentation that make the origin and quantities traceable at all times.
Executive Summary
The EUDR requires complete traceability of raw materials, including intermediate products. Companies must be able to prove the origin of each relevant raw material down to the geographical area of origin. The formal DDS submission is the responsibility of the initial supplier. Downstream actors must above all keep references and the quantity-related allocation consistent.
Collective batches and mixing processes require particular care: The composition, quantity ratios and origin of each partial quantity must be documented. A separate DDS is usually not required downstream if valid DDS references are available for the original batches. Internal processing steps do not trigger a new reporting obligation as long as traceability is maintained.
In practice, a risk-based approach is recommended: the higher the risk, for example due to country classification or data gaps, the more in-depth the documentation and checks need to be. Digital systems and automated protocols help to efficiently map even complex supply chains in compliance with EUDR.
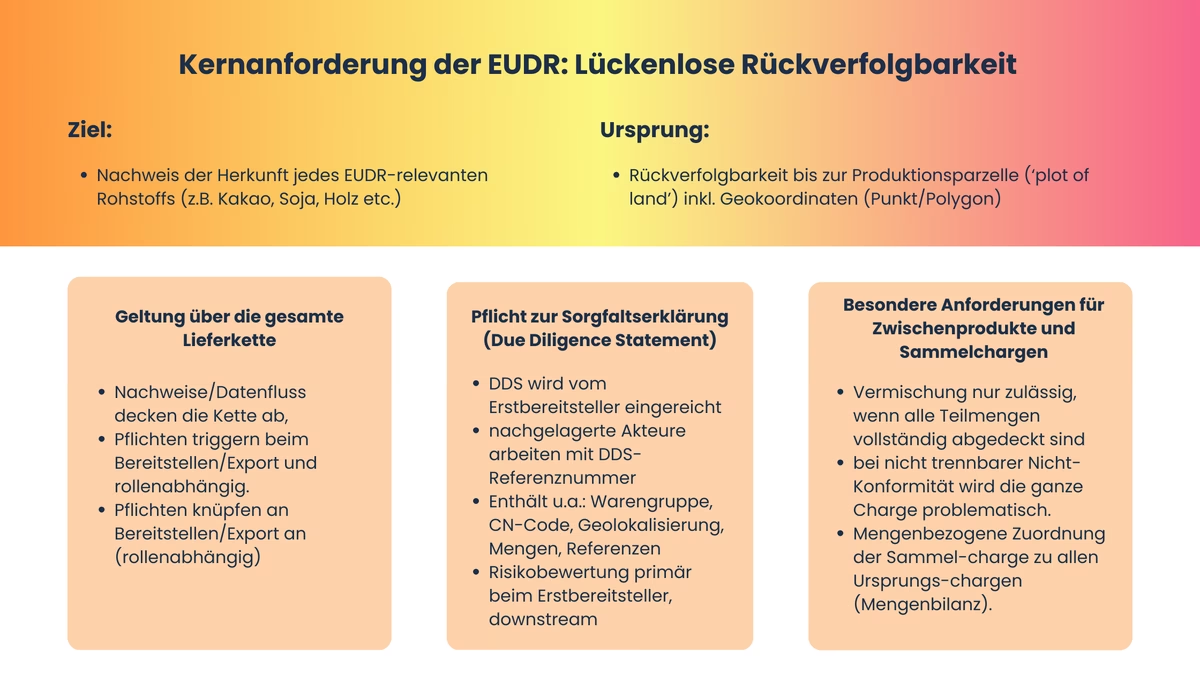
The EUDR basic requirement: Complete traceability
What does "full traceability" mean?
The EUDR (EU regulation against deforestation) requires companies to be able to prove exactly where the raw materials in certain products come from - this is what is meant by "full traceability". For every product covered by the regulation (for example cocoa, coffee, soy, palm oil, beef or wood), it must be clear from which farm, plantation or forest the raw materials originally came from. Traceability must be documented precisely, down to the geographical area, i.e. the place on the map where the raw materials were grown or harvested. 'Complete' means that each relevant batch can prove its origin back to the production parcel(s) - including the geolocation (point or polygon) of each area from which the raw materials originate. If a batch consists of several areas of origin, all areas must be indicated separately.
This traceability must apply throughout the entire supply chain: from cultivation to harvesting, transportation, processing and trade, whether inside or outside the EU. It is therefore not enough just to know who you bought the product from. You have to be able to trace it all the way back to the source.
A DDS is generally issued by the companies that supply or export a relevant product to the EU market for the first time. Downstream actors must accept, document and pass on the DDS reference numbers and remain jointly responsible for the conformity of the products they trade.
All intermediate steps must be documented so transparently that they can be traced down to area or parcel level. For intermediate products and collective batches in particular, this means that their composition and further processing can also be clearly documented.
Intermediate products are part of the supply chain
Processing raw materials into intermediate products is commonplace in many industries. For example, cocoa beans are processed into cocoa mass or butter, palm oil is split into fractions or wood assortments are transformed into semi-finished products (such as wooden boards, veneers or chipboard) before they are incorporated into the end product. From a legal perspective, intermediate products are an integral part of the supply chain. Legislation therefore also requires consistent proof of origin and processing for them.
However, processing stages that influence the end product (such as mixing, refining, portioning) increase the requirements for the clear batch and quantity balance, especially in the case of mixtures or collective batches. However, they do not trigger a new EUDR notification as long as the resulting relevant product is not made available on the Union market or exported. A DDS is generally submitted by the first supplier. As a rule, downstream actors do not have to submit a new DDS, but must take over and document the documents of the upstream declaration.
Documentation requirements for processing companies
What actually needs to be documented?
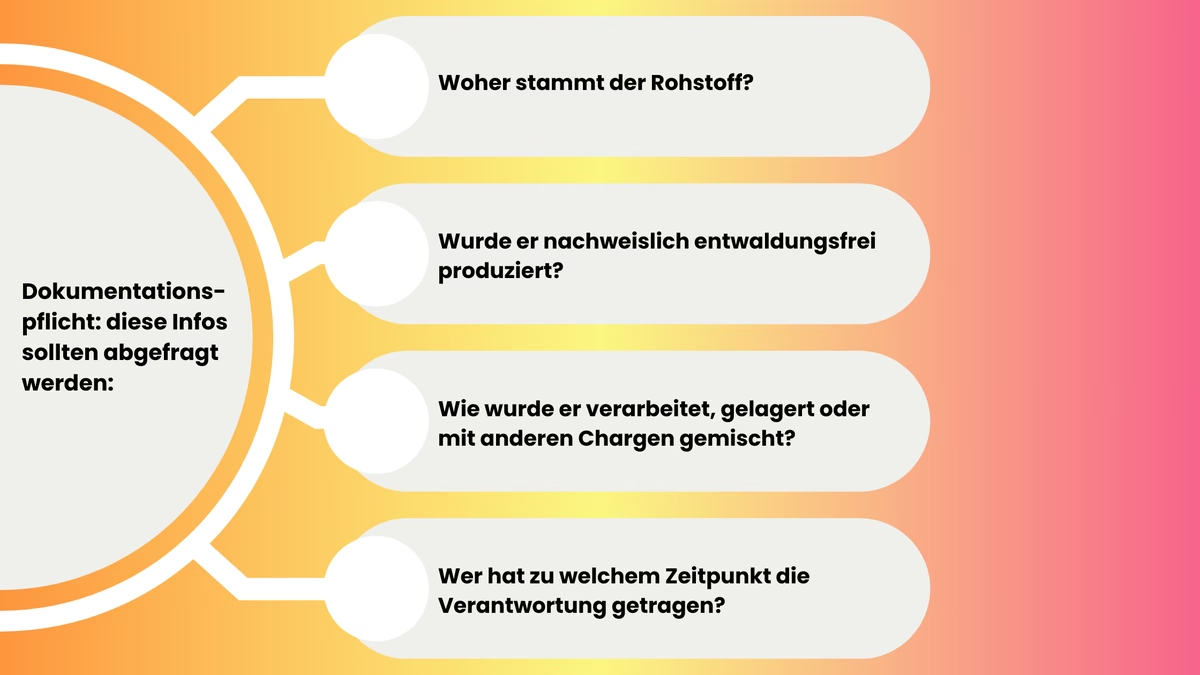
The question of what needs to be documented in detail for intermediate products or processed raw materials is best answered by looking at the objectives of the EUDR. The focus here is on an unbroken chain of relevant information:
- Where does the raw material come from?
- Was it demonstrably produced without deforestation?
- How was it processed, stored or mixed with other batches?
- Who was responsible and when?
- Which DDS reference number(s) cover the original batches used - and what role does the company (initial supplier vs. downstream actor) have with regard to due diligence and documentation?
For each intermediate product, it should therefore be documented exactly from which original batches it was produced. Clear batch identification, delivery documents and production records that reflect the number and type of raw materials used are generally sufficient. If several original batches are mixed, the exact (quantity-related) composition must be traceable. The challenge increases the more often collective batches or intermediate products from different sources of origin are formed, for example when processing large quantities of soy, cocoa nibs for chocolate production or sawmills that bundle logs from different parcels.
Particularly important and always required are details of the contacts entering the supply chain and the disclosure of relevant information (parcel ID, proof of origin, deforestation-free).
Companies that carry out further processing must document all plausible processing steps that are required for the consistency of the proof of origin and therefore for EUDR-compliant traceability. Pure transportation or storage movements do not trigger an additional EUDR notification (DDS). However, they must be documented if they are required for the batch and quantity balance, particularly in the case of splits, stock transfers or if stocks from several batches of origin are managed in parallel.
Practical tip: Documentation according to relevance and risk
In practice, a risk-based approach to documentation is recommended. Not every internal movement needs to be logged, as long as the origin and quantity relationship of primary materials remain secure and traceable. It is important to note that if a product has a known risk, for example because it originates from an area with a high risk of deforestation (high-risk country) or because the origin is difficult to trace (e.g. due to frequent mixing of different sources of origin), then particular care must be taken. In these cases, companies should deepen their documentation and - depending on their role (initial supplier vs. downstream actor) - either tighten up their own risk analysis or at least ensure additional plausibility/red flag checks and a stricter batch and quantity balance.
For example, a soy company that processes soy from different countries of origin into soybean oil can use clear labeling and digital tracking of input batches to ensure that all downstream production is correctly allocated. This can reduce risks during audits. At the same time, weak points in traceability can be better traced and the legal requirements of the EUDR for collective batches and intermediate products can be fulfilled in a compliant manner.
Handling collective batches and stock goods
The challenge of mixing
Especially when bundling large quantities of goods, whether soy, palm oil, coffee or wood, raw materials from different plantations or countries are brought together to form a so-called collective batch. These collective batches pose a particular challenge from the perspective of the EUDR, as precise traceability back to the origin must be guaranteed for each partial quantity. In practice, this often conflicts with the legal text: Technically, the origins represented in a collective batch can span dozens, sometimes even hundreds of individual producers. Mixing is permitted as long as all partial quantities are fully georeferenced and compliant. A mass balance chain that offsets compliant with unknown or non-compliant goods is not sufficient according to the EUDR.
Nevertheless, the EUDR requires that the consolidated batch can be assigned to the underlying batches of origin (including geolocalization) in terms of quantity. Companies are therefore faced with the task of documenting incoming goods and relevant stock movements in such a way that the DDS reference(s), original batches and quantity balance remain consistent without each movement triggering a new DDS.
Digital warehouse management systems, detailed delivery bills and production records play a key role here. If further processing, such as grinding, extraction or portioning, takes place at a later stage, the composition of the original data must be fundamentally retained. As soon as a proportion is not compliant or cannot be clearly assigned, the entire collective batch becomes risky or potentially non-compliant, which is why separation and clear quantity logic are crucial.
What is permitted and what must be documented?
The EUDR permits additions and mixtures as long as traceability is guaranteed for each partial quantity of the collective batch. It is therefore still permitted to form collective batches and process them further if a transparent system ensures that the origin and quantity ratio are maintained and - depending on the role - either the due diligence assessment of the initial supplier is traceable or downstream actors document red flag checks and a consistent quantity balance. This is common in the cocoa sector, for example, where individual export batches originate from several farmer cooperatives, but each partial batch can be documented back to the community of origin.
In storage practice, the following applies: Mixed batches are EUDR-compliant if their origin can be precisely documented and the risk assessment explicitly addresses the mixing. Commingling is permitted as long as each part of the consolidated batch can be documented with geolocation and mass balance. A mass balance logic that 'covers up' untraceable quantities is not sufficient.
This requires posting withdrawals and processing steps in such a way that the quantity-related composition remains traceable at all times (especially in the case of splits, consolidations and new mixtures). Companies can fulfill this requirement by keeping an accompanying overview for each collective batch, which bundles all relevant origin, quantity and storage information. This should also be the case for longer storage periods and frequent transfers, as long as there is no subsequent mixing with new, untraceable quantities.
Practical experience from the timber industry shows: When companies use barcoded storage units, digital booking systems and automatic production logs, traceability becomes much simpler and more transparent. The manual effort required for verification is significantly reduced. At the same time, even complex collective batches can be documented reliably and in compliance with EUDR.

Processing steps and "new placing on the market"
When does an intermediate product become a new EUDR case?
One of the most common uncertainties in relation to EUDR traceability is when a separate EUDR notification is required for an intermediate product or a new processing step.
The core of the regulation is: If a product is made available through a change or is taken over by another market participant, a new due diligence declaration is required. It is important to note whether the product has been manufactured exclusively from products that have already been subject to due diligence. If this is the case, it is possible to refer to previous DDSs. However, this does not exempt from the responsibility for the conformity of the products.
Example: A company imports cocoa beans from outside the EU and processes them into cocoa liquor. Even if the cocoa mass remains in the possession of the same company within the EU, it is only used for its own needs and is not passed on to third parties, a DDS must be created.
As the first distributor on the Union market, the company has to fulfil the full due diligence obligation and submit a due diligence declaration with the geolocation data of the cocoa beans (HS 1801) in the information system. For example, if the cocoa mass (HS 1803) is sold to a chocolate factory, the factory converts a relevant product into other relevant products and is therefore considered a downstream non-SME market participant for chocolate (HS 1806).
The chocolate factory as downstream operator must adopt, check the plausibility of and document the reference numbers of the original DDS, but does not necessarily have to submit a new DDS (provided that all inputs are already covered by a DDS/simplified declaration). Since the chocolate was produced using the cocoa beans already subject to due diligence (HS 1801), the factory can refer to the due diligence declaration submitted by the cocoa bean buyer, stating the corresponding reference number. However, it must first ensure that the upstream due diligence obligation in accordance with the EUDR has been fulfilled. The processor remains responsible for the conformity of the products concerned.
An intermediate product becomes a new EUDR case if it is not fully covered by an existing DDS/simplified declaration (e.g. new, non-covered inputs, mixing with unknown origin, missing references) - then the actor becomes 'first placer' again and must do full due diligence + DDS.
Transforming processing steps (e.g. grinding, refining, extracting, fractionating) without transfer of ownership do not in themselves trigger a new EUDR notification, but the provision to new legal entities does. In this case, the downstream obligation begins for the receiving company to ensure full EUDR traceability based on the origin data and quantities provided.
Exemplary processing chain
Let's assume that a Brazilian farmer delivers soybeans to a regional collection warehouse. Beans from 30 other farms are collected there and cleaned together. The exporter forms a collective batch from this, which is sent to the EU and documented via an EUDR declaration. An EU importer sells part of the collective batch to an oil mill that presses soybean oil. As long as the oil mill stores and processes the oil, it can rely on the documentation of the original collection batch.
If it passes the oil on to a food manufacturer, the manufacturer (depending on its role as first downstream/downstream operator) must adopt the DDS reference of the upstream declaration and document its quantity/batch allocation in such a way that traceability is maintained. As a rule, a separate DDS is only required if he himself acts as the first supplier or if the upstream cover/references are missing. Whether margarine is EUDR-relevant here depends on the specific CN/HS code and whether it is listed in Annex I. If the code is not listed, the product is not EUDR-relevant (even if it contains soy/palm oil).
To ensure that the supply chain remains EUDR-compliant, at least the following information must be traceable along the chain (depending on the role):
- Parcel IDs of the original farms
- Quantity ratios (for example, what proportion of the collection batch comes from which farm)
- Complete transfer documentation (production, warehouse and delivery bills)
- Assessment and documentation of any risks (such as mixing ratios, known origin from high-risk areas, etc.)
At no point may the transparency of the origin of the respective partial quantity be lost, otherwise the end product loses its EUDR conformity. A non-compliant part of a product must be identified and separated from the rest before it is placed on the EU market or exported. This part may not be placed on the EU market or exported. If identification and separation is not possible because the products have been mixed with the rest, the entire product is non-compliant. It may not be placed on the EU market or exported.

Recommendations for action in practice
Establish clear processes and systems
The most important success factor for trouble-free documentation along complex supply chains lies in structured, clearly defined processes. Companies should record all relevant processing steps, transfers of ownership and quantities as well as mixtures in a standardized system, for example using ERP systems with integrated traceability modules or software solutions such as the lawcode Suite. In larger organizations, a clear assignment of roles and responsibilities is recommended to ensure that all internal and external transfers are mapped in compliance with the EUDR.
The standardized recording and transfer of parcel IDs, proof of origin and processed quantities is also essential in order to ensure complete EUDR traceability within the shortest possible time, even in the event of inspections or audits. Companies that have so far mainly used paper-based documentation should consider switching to digital processes in order to improve efficiency, accuracy and legal certainty in the long term.
Managing interfaces - internally and externally
An often underestimated risk factor in EUDR documentation practice is interface management. Whenever material flows cross internal departmental, plant or site boundaries or change between different supply chain actors, the risk of information loss, transmission errors or ambiguities with regard to source data increases. Companies should ensure that all relevant interfaces are secured through consistent data transfers, clearly defined handover processes and regular control mechanisms.
Collaboration with external partners, for example in contract processing, warehousing or external transport, must also be designed in such a way that all relevant EUDR obligations are fulfilled seamlessly. It is advisable to contractually define which information obligations apply, e.g. with regard to the transmission of digital source data in the agreed format. The use of industry standards to ensure seamless traceability in practice should also be agreed in advance.
Allow risk-based documentation
The EUDR does not require a complete individual record of all internal steps; instead, a risk-based documentation approach can be chosen. This means that companies can rely on simplified documentation methods where the origin of the material remains clear and traceable without risk factors. Only when risk factors are added, such as repeated blending, additions from non-certified sources or processing from areas with an increased risk of deforestation, must the documentation be supplemented with additional evidence, protocols and risk assessments. Depending on the role (primary supplier vs. downstream actor), risk-based means either an in-depth risk analysis (primary supplier) or stricter plausibility/red flag checks plus a reliable batch and quantity balance (downstream).
Practical experience, particularly from the cocoa and palm oil sectors, shows that risk-based documentation is not only legal, but also audit-proof, provided that internal company instructions, digital protocols and traceable risk assessments are used systematically. As a result, companies benefit from greater efficiency without breaching the EUDR collective batch and intermediate product regulations.
To Do:
- Record all processing steps, quantity transitions and blending in a standardized system, ideally digitally with clear roles and responsibilities.
- Secure all internal and external interfaces through consistent data transfers and clear handover processes and contractually define information obligations with external partners.
- Use a risk-based documentation approach: Simplify where the origin is clear and deepen evidence and risk checks specifically where there are mixtures, data gaps or high-risk areas.
Conclusion
The EUDR does not require complete archiving of every internal intermediate step. The decisive factor is that the origin, composition and quantity ratios can be clearly and verifiably retrieved at any time. Collective batches and intermediate products are not an obstacle as long as the quantity-related allocation to the original batches is maintained.
The role logic is clear: the initial provider submits the formal declaration of due diligence, downstream companies keep references and quantity balances consistent, with in-depth checks where risks or an increased country classification require it. The postponed application dates until the end of 2026 offer the opportunity to standardize processes and digitalize data flows. So that EUDR compliance is not only achievable, but permanently manageable.
Frequently asked questions
In practice, an intermediate product is understood to be all processing stages between the raw material and the end product. However, only those intermediate or end products that are listed as relevant products in Annex I (e.g. cocoa mass/butter for cocoa, soybean oil/bean meal for soy, sawn timber/panels for wood - depending on the CN/HS code) are relevant to the EUDR. Products outside Annex I are not covered by the EUDR, even if they were manufactured from an EUDR raw material.
Not every action needs to be logged individually. It is crucial that your internal documentation reliably maintains the batch and quantity logic as well as the assignment to the areas of origin (geolocalization/plot of land). A new EUDR notification or DDS is not triggered by processing, but typically only when a relevant product is made available on the Union market or exported by the actor acting as the first supplier.
Collective batches are permitted, but you need a quantity-based allocation of the collective batch to all original batches used. In practice, this means
- Geolocalization up to the production parcel ("plot of land") for all shares (point/polygon),
- Quantity ratios/batch mapping (which proportion comes from which original batch(es)/area),
- Records that make the composition traceable even after mixtures or withdrawals. If a proportion cannot be clearly assigned or is not compliant and cannot be separated, the collective batch as a whole becomes problematic.
Companies should rely on process-oriented, digital systems that structure input, processing and output data and link them in a traceable manner. Employees should also be trained and clear responsibilities established. In this way, all EUDR traceability requirements can be reliably met, even for complex collective batches and intermediate products.

Matthias Klein
LinkedInESG-Compliance Experte · lawcode GmbH
Matthias Klein berät Unternehmen bei der Umsetzung von Supply Chain Gesetzen wie der CSDDD und begleitet die Implementierung digitaler Lösungen für rechtssichere Lieferketten. Seine Fachbeiträge auf dem lawcode Blog verbinden regulatorische Tiefe mit praxisnahen Handlungsempfehlungen.





