Important facts
- What is a due diligence statement (DDS)?
- A DDS is a mandatory due diligence declaration that proves that a product is deforestation-free, based on a clearly defined product batch with all relevant information on origin, quantities and risks.
- How does the practical implementation of a DDS work?
- All information about a batch, such as the parcel of origin, quantities, processing steps and transportation routes, is collected, checked and submitted digitally as a due diligence declaration.
- What does the duty of care relate to - the batch or the delivery?
- The DDS always refers to the batch. Deliveries are merely the physical movement of goods, while traceability and documentation are based on the defined batch.
- What needs to be documented?
- The parcel of origin, geographical data, quantities, supply chain, transportation and processing steps as well as risk assessments, including mixing or repackaging, must be documented.
- What happens if the DDS documentation is incorrect or incomplete?
- Missing or incorrect information can lead to sanctions, fines or exclusion from the EU market.
Executive Summary
From December 30, 2026, companies that place EUDR-relevant products on the EU market or export them must present a DDS in the EU information system, either as a separate declaration or by referencing an existing DDS. The DDS relates to a defined product quantity and can cover several partial deliveries if the origin data remains the same, but must be available before the first delivery.
The central requirement is the clear identification of batches with complete documentation of origin, quantity, transportation and processing. Traceability must not be interrupted in collective warehouses and mixtures; all origins must remain clearly assignable. Mixing with goods of unknown origin is not permitted.
Missing or incorrect documentation can lead to sanctions and market bans. Clear batch allocation, consistent use of reference numbers and the training of all departments involved are decisive success factors for legally compliant EUDR implementation.
What is the Due Diligence Statement (DDS) and why is it important?
Overview of the DDS obligations of the EUDR
The DDS is the central instrument of the EUDR. Depending on the size of the company, the obligations will apply from the end of 2026 or mid-2027 to all companies that supply or export deforestation-relevant raw materials such as wood, cocoa, coffee, rubber, palm oil, beef or soy to the EU market for the first time.
The prerequisite is a robust due diligence system that identifies, assesses and documents risks along the supply chain. The core is complete traceability down to the production parcel: companies must record geolocation data, quantities, delivery routes and actors involved. The goods must be deforestation-free, with a deadline of December 31, 2020, and comply with the legal requirements of the country of production.
The final step is the electronic submission of the DDS: formal confirmation that information is available, a risk analysis has been carried out and, if necessary, risk reduction measures have been implemented. Without a valid DDS, a product may neither be sold in the EU nor exported.
Important: Not every role in the supply chain is automatically subject to the same level of obligation. Simplifications are provided for downstream market participants and many SMEs in particular, but the obligation to keep information in a traceable manner, pass on references and organize your own supply chain in such a way that the DDS logic works reliably in practice remains.
DDS goal: No deforestation via products in the EU
With the due diligence obligation, the European Union wants to prevent products from being imported or exported that are associated with deforestation or the destruction of forests. The EUDR due diligence declaration is the central link between the documented supply flows and the risk-oriented rejection or approval by the authorities. This not only protects consumers, but also creates new requirements and responsibilities for trade, industry and the entire upstream value chain.
Importance of documentation for all parties involved in the supply chain
The scope of the verification and documentation obligations extends to everyone involved in the supply chain. Without complete documentation on the place of origin, quantity, delivery route and transformation processes in the company, there is not only the threat of sanctions, but also the loss of market participation in the EU.
Practical experience shows that around 80% of companies with relevant raw material use report difficulties with traceability and collecting information on origin, as studies by industry associations confirm.
It is clear that a strong and well-functioning DDS infrastructure is very important - not only for compliance with regulations, but also for a sustainable and future-proof orientation of the company.
DDS and the reference to batch and delivery - how is this regulated?
Information and definitions for practical use
For a successful and smooth implementation of the EUDR, it is crucial to use key terms consistently within the company and to make a clear distinction between legal logic and operational practice. Two terms come up particularly frequently in day-to-day implementation: "batch" and "delivery". Both are important, but do not mean the same thing.
In practice, "batch" usually refers to an internally defined, clearly definable quantity of goods, i.e. a batch or lot logic with which companies bundle products under comparable manufacturing, processing or procurement conditions. It is important to note that "batch" is primarily an operational control term. It helps to structure flows of goods in such a way that all relevant information and evidence can be reliably assigned, such as origin, production area/geolocation, actors involved, quantities, processing steps and documents for traceability. It is precisely this unambiguous allocation that is the key later on when information needs to be consolidated internally or documented in a traceable manner for business partners and authorities.
This is to be distinguished from "delivery". A delivery describes the actual transportation or physical handover of goods to the next player in the supply chain. It is typically accompanied by documents such as delivery bills and shipping documents and documented in the ERP system as a goods issue or goods receipt. The decisive factor is that a batch can extend over several deliveries and, conversely, a single delivery can contain several batches. This flexibility is normal and is common practice in many industries.
Batch = internally defined, definable quantity of goods for the structured allocation of origin, quantities and proofs.
Delivery = physical movement of goods to the next actor. A batch can comprise several deliveries - and vice versa.
For EUDR implementation, it is therefore less about the term "batch" itself, and more about the clear delimitability and traceability of the goods that are placed on the market or exported. In practice, this means that companies should set up their batch and delivery logic in such a way that the relevant products and quantities can be traced at all times, including the linked origin and traceability data. Clean documentation and clear handling of these terms not only help to implement EUDR requirements consistently. They also improve transparency within the company, reduce frictional losses between purchasing, logistics, quality and compliance and make it much easier to identify risks at an early stage and minimize them effectively.

Basic rule of the regulation: DDS refers to a defined quantity of goods, not to the delivery
The logic of the EUDR-DDS is not linked to individual delivery bills, but to a clearly defined quantity of relevant products that are to be placed on the EU market or exported. For this defined quantity, market participants must collect all necessary information and evidence, carry out a risk analysis and, if necessary, implement risk mitigation measures. The DDS is therefore formal proof that the due diligence obligations have been fulfilled prior to market access.
In practice, deliveries are merely the physical movement of goods; they can represent the underlying quantity in one or more shipments. It is therefore important to note that a DDS can cover several partial deliveries, but must be submitted before the first delivery. And it is only valid for the quantity specified in the DDS: as soon as this quantity has been fully placed on the market or exported, a new DDS is required for additional quantities. In practice, a DDS should also not be used "in stock" over long periods of time, but should remain traceable in terms of time and quantity.
A practical example: An importer brings 20 tons of cocoa into the EU. For this defined quantity, all origin data (incl. production areas/geolocation), proofs and risk assessments are collected and merged in a DDS. Whether the 20 tons arrive in one shipment or in several partial shipments is of secondary importance for the DDS logic. The decisive factor is that the DDS is available before the first partial delivery and that the quantities actually placed can be clearly traced back to the quantity declared in the DDS at any time.
Relevance for reference numbers and product labeling
Well-maintained and clearly assigned reference and verification numbers from the EU information system are essential for a stringent DDS system. They enable DDSs to be consistently referenced along the supply chain and to be clearly linked internally with batch/lot IDs and delivery or consignment numbers. This means that the flow of goods remains traceable at all times, from a compliance perspective as well as for recalls or spot checks.
In practice, these links should be consistently maintained in the ERP and in the documentation (e.g. delivery bills, invoices, incoming/outgoing goods) in order to avoid media disruptions and allocation errors. Modern ERP solutions and traceability tools support this with automated workflows, versioning and audit-proof filing. Standardized product and packaging labeling (e.g. barcode/QR/RFID) is not mandatory, but can simplify data collection, reduce errors and significantly speed up checks and audits.
How do you deal with collective batches and stock items?
Problem: Lack of separation or mixing in the warehouse
In practice, companies often face organizational and technical challenges when storing raw materials and products, especially when goods are kept as bulk stocks or in loose form in silos, tanks or storage boxes. In such set-ups, different batches of goods can quickly become mixed or cross-contaminated. This becomes critical at the latest when partial quantities are removed and it is no longer possible to reliably trace the origins of these quantities afterwards.
Typical examples are import and storage processes for grain, where harvests from different regions are brought together, or for oilseeds and other bulk goods, where batches are mixed during operation. Without clear separation logic and reliable booking rules, there is a risk that information on origin, quantity references and proofs can no longer be clearly assigned.
This is precisely where the EUDR comes in with its high requirements for traceability and verifiability: the decisive factor is not that physical mixing is excluded in every case, but that mixing does not result in the relevant quantities losing their clear assignment to origin and geolocation. Models in which goods of unknown origin or with a different risk profile "run along" mathematically (mass balance logic) are not permitted.
For companies, this means a noticeable effort: they must design their warehouse and process logic in such a way that even with bulk storage, it remains traceable which origins and quantities are contained in a definable quantity of goods and that the documentation remains auditable at all times. This usually requires a combination of organizational rules (e.g. clear lot definitions, blocking and release processes, defined mixing points, responsibilities) and technical measures (e.g. WMS/ERP workflows, clear references, digital verification chains). Equally important are training courses for warehouse staff and clear instructions on how to deal with decanting, mixing, residual quantities and deviations.
To summarize: Companies are required to proactively set up their warehousing in such a way that traceability also works under real conditions. With clean processes, consistent documentation and suitable systems, legal risks can be significantly reduced and transparency, efficiency and trust along the supply chain strengthened at the same time.

Do separate DDS documents have to be created for each batch?
The EUDR does not require a blanket due diligence declaration "for the entire warehouse", but a DDS for clearly defined relevant goods before they are placed on the market, made available or exported in the EU.
In practice, this means that the DDS must always refer to a clearly defined consignment or lot. If goods from different origins are combined (e.g. a coffee blend from several regions), a new relevant item is created for which a corresponding DDS must be submitted - including the geolocation data of all underlying production areas. Models in which compliant goods are "mixed" with goods of unknown origin using mass balance/percentage logic are not permitted. At the same time, information from previously submitted DDSs can be referenced upstream for a new DDS so that data does not have to be entered twice unnecessarily.
Possibilities of implementation in practice
Various implementation options are widely used to avoid errors or bottlenecks. Many companies use digital merchandise management and track & trace systems that automatically check whether each withdrawal, delivery or reworking is assigned to a batch. In larger agricultural businesses or commodity traders, color and storage zone concepts, barcode scans and differentiated storage areas are also in use. At an operational level, every company should define internal work instructions that regulate how to proceed when combining, portioning or repackaging DDS-relevant material.
In practice, there are often mixed approaches. Certain mixtures are managed as separate sub-batches as long as it is clearly documented where the individual ingredients come from. Regular internal audits and training of the responsible employees are crucial in order to implement the due diligence obligations effectively and with as few errors as possible.
Special cases and common pitfalls
Different suppliers for the same products
A particularly challenging situation arises when a company purchases the same types of goods from different suppliers, such as soybeans from several sources. In these cases, the documentation and control effort increases significantly because the origin, production areas (including geolocation data), quantities and proofs must be reliably assigned to each origin.
It is important to note that the EUDR does not necessarily require "one DDS per supplier" or "one DDS per delivery", but that all relevant products and quantities that are placed on the market or exported are fully covered by a DDS and that the due diligence for these quantities has been demonstrably carried out.
At the same time, it is not permissible to combine goods of different origins in such a way that the clear allocation is lost or shares of unknown or unsecured origin "run along" mathematically. If mixing takes place later in the warehouse, it must therefore remain clear which origins and quantities are contained in the definable quantity of goods. For the company, this means that end-to-end transparency must be ensured, both organizationally and in terms of the system, as early as the goods receipt posting and throughout all further processing and delivery processes.
Repackaging or order picking in the warehouse
In logistics practice, goods are often repacked or picked for individual customers. This raises the question of how the DDS documentation must be maintained if the flow of goods no longer corresponds to the original layout of the stock. It is crucial that the chain of evidence and traceability is maintained even after repackaging, partial removal or reassembly: The definable quantities must still be able to be clearly traced back to the underlying origins (incl. geolocation data) and the corresponding DDS references.
In practice, this means that although repackaging and picking change the physical flow of goods, the documentation must remain consistent, especially where goods are later placed on the market, made available or exported as a relevant product. Many companies solve this using clear ERP/WMS rules and digital links by carrying the reference and verification numbers from the EU information system as "anchors" in their inventory and movement data. Serialized identification (e.g. QR code, barcode or RFID) is not mandatory, but can help to avoid media disruptions and significantly speed up checks, audits or spot checks.
Practical example: Loose stored material with individual delivery
An illustrative example from everyday life: A retailer stores rubber in several silos, some of which have different origins and each with assigned proofs. The rubber is delivered to the processing industry in partial quantities or as mixtures, depending on customer requirements. It is crucial that the company can document the origins (incl. production areas and geolocation) and which quantities are contained in the respective delivered, definable quantity of goods at all times, even in bulk storage. Mixing must not lead to a loss of origin and traceability or to goods of unknown origin being "included" in the calculation (mass balance logic).
If new, definable quantities are created through merging or withdrawal processes that are to be placed on the market or exported as a relevant product, it must be ensured that these quantities are fully covered by a DDS, including the relevant origin data. Robust ERP/WMS posting rules and measurement data (e.g. weighing and fill level checks) provide particular technical support here in order to make stocks, withdrawals and mixing points verifiable, especially in the case of high throughput and complex storage processes.
The most important thing for loose storage:
→ Each delivered quantity must be clearly traceable to its origins and geolocation data
→ Mixing must not lead to loss of traceability, goods of unknown origin must not "run along"
→ New definable quantities must be fully covered by a DDS
→ ERP/WMS booking rules and measurement data are central technical aids
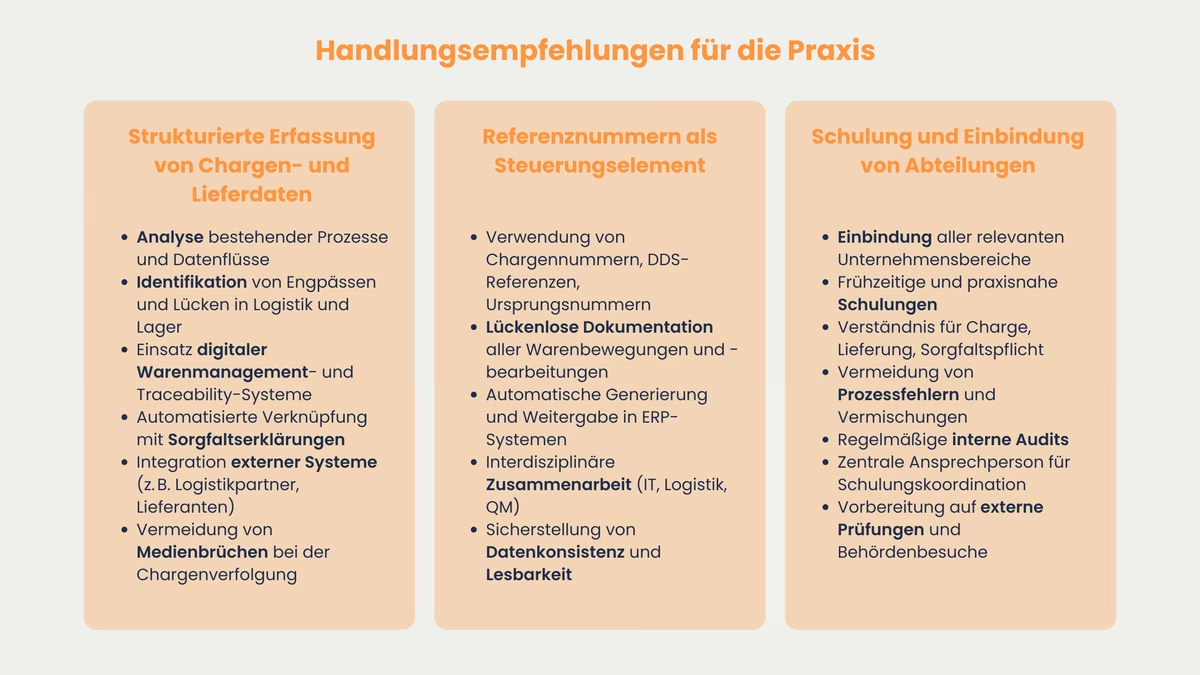
Recommendations for action in practice
Establishment of a structured recording of batch and delivery data
The most important basis for a smooth and legally compliant EUDR implementation is the establishment of a structured system for recording all batch and delivery-relevant data. Companies should analyze existing processes at an early stage, identify bottlenecks in the data flow and close gaps in warehouse or production logistics. Digital goods management and traceability systems offer a way of automatically linking all incoming goods, internal stock transfers and deliveries with the associated due diligence declarations. It is also advisable to integrate third-party systems, such as those of external logistics partners or suppliers, into your own database at an early stage in order to avoid media discontinuities in batch tracking.
Use of reference numbers as a central control element
The consistent use of reference numbers (batch number, DDS reference, origin number) not only increases transparency, but is also a central control element in the context of the EUDR-DDS. Every movement of goods, every storage event and every processing operation should be documented with these references, from goods receipt to internal processing and delivery to the end customer. Modern ERP systems enable these reference numbers to be automatically generated, maintained and forwarded to downstream systems. Specialist departments such as IT, logistics and quality management should work closely together to ensure data consistency and legibility, even with complex product ranges and multi-level supply chains.
Training and involvement of relevant departments
The due diligence obligations have a deep impact on several areas of the company, from procurement, production and warehousing to sales and the legal department. Early and continuous training of all employees who come into contact with the processes is crucial. Only those who understand the logical link between batch, delivery and due diligence declaration can avoid process errors, unauthorized mix-ups or gaps in the documentation. Practical training courses based on typical company processes, regular "incoming goods inspections" through internal audit series and a central contact person who answers questions and coordinates training courses are helpful. A comprehensively trained workforce is a decisive competitive advantage, particularly with regard to inspection visits by authorities or external inspection bodies.
Conclusion
The clear insight from the EUDR obligations: The DDS is always related to the batch, the physical delivery only acts as a subsequent event. The clean separation and documentation of batches is therefore the backbone of EUDR compliance. Companies that implement this consistently avoid serious risks and create a solid foundation for sustainable, future-proof supply chains. Only complete, tamper-proof and transparent documentation of all batches and associated deliveries can minimize the risk of compliance violations. With its strict requirements for due diligence declarations, the EUDR does involve a lot of effort. However, the benefits, such as legal certainty, greater customer trust and sustainable positioning, pay off in the end. In times of increasing market surveillance and social responsibility, this is indispensable.
Companies wishing to take a closer look at the implementation of the EUDR, for example the technical implementation of batch tracing or the establishment of internal processes, should read the official guidelines of the EU Commission. Expert forums and industry-specific recommendations from the respective associations also offer valuable support.
Frequently asked questions
An EUDR batch is a defined quantity of a product that was obtained, processed or manufactured under the same conditions. It is also clearly identifiable on the basis of unique parameters (date, parcel of origin, production unit). In the context of EUDR, each batch is assigned its own declaration of due diligence in order to be able to provide precise proof of origin and risk profile.
The EUDR declaration of due diligence is drawn up for each relevant batch. It contains detailed information on the country of origin, the parcel, the harvest date, the delivery route and all processed quantities. The declaration is passed on to the responsible authorities and business partners in digital form and forms the basis for inspections by the authorities and internal audits. In most cases, the due diligence declaration is integrated into the company's ERP system and maintained automatically.
Yes, several deliveries can reference a single batch. It is crucial that the declaration of due diligence always remains batch-specific. Partial deliveries, distribution in several portions or internal stock transfers must be documented in the systems in such a way that the reference to the original batch DDS remains visible at all times.
As soon as different batches are mixed in the warehouse, the company must create a new, so-called collective batch. A separate DDS must be created for this new batch, whereby the respective quantity shares and origin information must be stored transparently and verifiably. Simply updating existing due diligence declarations is not permitted.
Incomplete documentation or incorrect allocation of batches can result in severe sanctions, fines and, in the worst case, exclusion from the EU market. In addition, reputational damage and additional operational costs arise from subsequent investigations and corrections. Stringent, early preparation is therefore essential for sustainable implementation of the EUDR.

Matthias Klein
LinkedInESG-Compliance Experte · lawcode GmbH
Matthias Klein berät Unternehmen bei der Umsetzung von Supply Chain Gesetzen wie der CSDDD und begleitet die Implementierung digitaler Lösungen für rechtssichere Lieferketten. Seine Fachbeiträge auf dem lawcode Blog verbinden regulatorische Tiefe mit praxisnahen Handlungsempfehlungen.